In this blog article, we will examine a few hypothetical scenarios that show the necessity of taking corrective action in several business areas. We’ll also talk about potential tactics and methods that could be used to stop or lessen the consequences of these hypothetical outcomes. […]
Quality Management
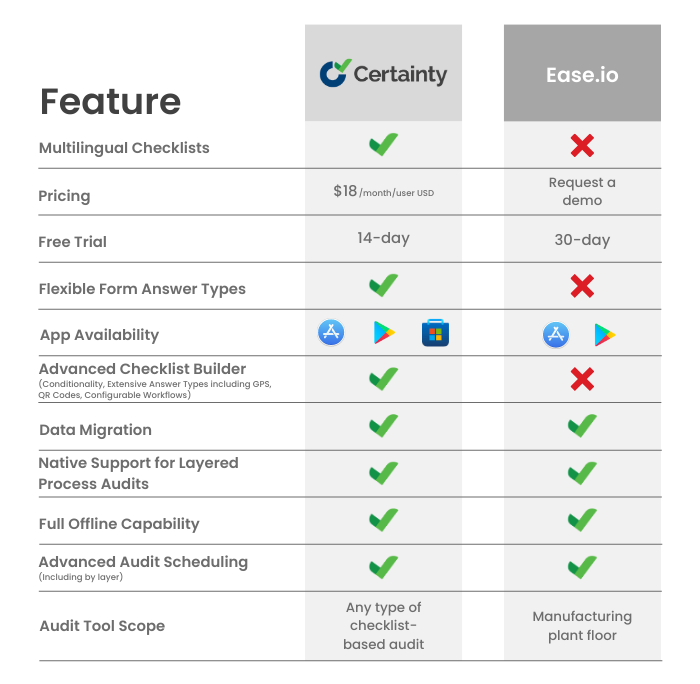
Ease.io vs. Certainty Software: Which Audit and Inspection Management Software is Right for You?
Overview If you are in charge of carrying out audits and inspections for your company, you are aware of how crucial it is to have dependable and effective software to handle your procedures. We at Certainty Software are aware that you want a tool that enables you to make and distribute checklists, gather and analyze […]

What is a BRC Audit and How to Perform One
Summary: A BRC (BRCGS) audit evaluates whether a food, packaging, or storage facility meets the British Retail Consortium's Global Standards for safety, quality, and operational compliance. Passing a BRC audit demonstrates to retailers and supply chain partners that your organization maintains rigorous controls over product safety, traceability, and continuous improvement. Structured preparation — including gap […]

SMED: Streamlining the Lean Manufacturing Process
Summary: Single-Minute Exchange of Die (SMED) is a Lean manufacturing methodology that reduces equipment changeover times to under ten minutes, dramatically increasing production flexibility and throughput. By separating internal and external setup activities and systematically converting or streamlining each step, SMED minimizes downtime losses that directly impact OEE, first pass yield, and cost of poor […]

Certificate of Conformity: The Ultimate Guide for Manufacturing Companies
Summary: A Certificate of Conformity (CoC) is an official document that verifies a product meets all specified quality, safety, and regulatory requirements before it enters the market or crosses borders. For manufacturers operating under ISO 9001, IATF 16949, or other regulatory frameworks, maintaining accurate and traceable CoC documentation is essential for audit readiness, customer satisfaction, […]

OEE Meaning in Manufacturing: How to Optimize Overall Equipment Efficiency
Summary: Overall Equipment Effectiveness (OEE) is a manufacturing metric that measures the percentage of planned production time that is truly productive, combining availability, performance, and quality into a single score. The average manufacturing plant operates at only 60% OEE, meaning 40% of capacity is lost to breakdowns, slowdowns, and defects. Improving OEE directly reduces the […]

Unlocking Efficiency: A Guide to Identifying and Eliminating Muda in Your Business Processes
Summary: Muda refers to any activity or process that consumes resources without adding value to the customer, and eliminating it is a core principle of Lean manufacturing and quality management. The seven types of Muda — overproduction, waiting, transport, overprocessing, inventory, motion, and defects — directly impact quality KPIs like first pass yield, cost of […]

Poka Yoke – The Ultimate Guide to Error Prevention
Summary: Poka Yoke (mistake-proofing) is a Lean manufacturing technique that designs processes and systems to prevent human errors before they occur, rather than relying on detection after the fact. Originally developed within the Toyota Production System, mistake-proofing devices and methods are now essential tools for reducing defect rates, improving first pass yield, and lowering the […]
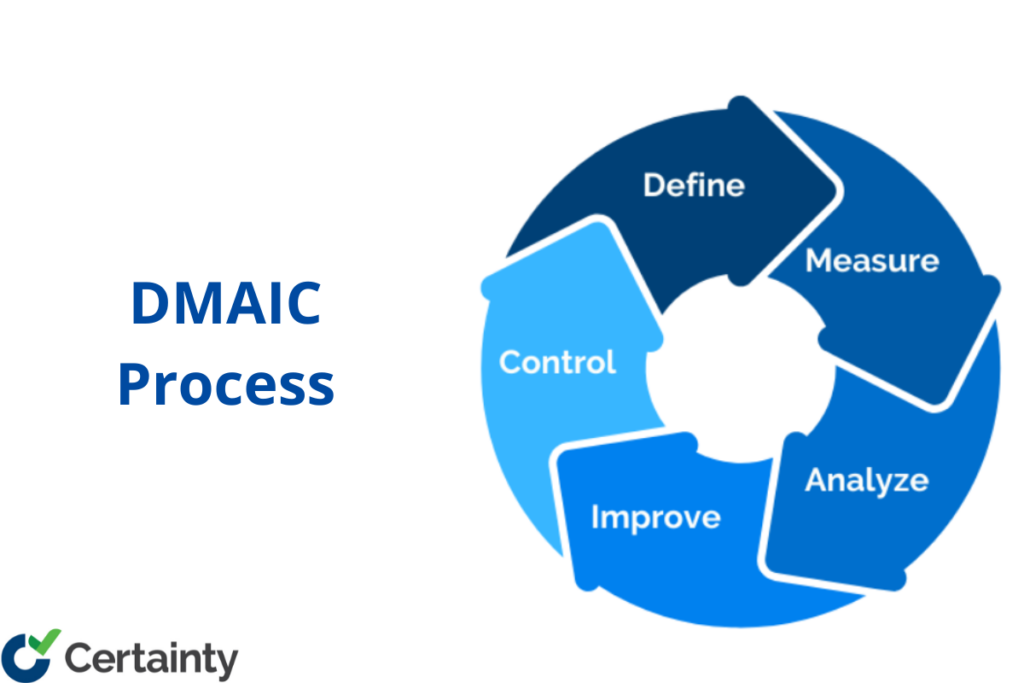
Implementing the DMAIC Process: A Comprehensive Guide
Summary: DMAIC (Define, Measure, Analyze, Improve, Control) is a data-driven process improvement methodology rooted in Six Sigma that helps organizations systematically reduce defects, variability, and waste. Each phase builds on the previous one, using statistical tools and quality metrics like first pass yield and cost of poor quality to guide decision-making. Implementing DMAIC within your […]

The Power of Integrated Risk Management: Enhancing Organizational Success
Do you want to learn how to manage risks effectively and efficiently in today’s complex and dynamic business environment? In this blog post, we will show you how integrated risk management (IRM) can help you achieve organizational success. We will also explain how Certainty Software can support your IRM initiatives. Read on to find out more. […]

8 Effective Risk Management Techniques for Today’s Challenges
From Bowtie Analysis, to training programs, discover 8 of the most effective risk management techniques and strategies your operations can implement quickly. Learn to how to better manage your risk mitigation efforts with this blog post. […]

Mastering 8D Problem Solving: A Comprehensive Guide for Businesses
Summary: The 8D (Eight Disciplines) problem-solving methodology is a structured, team-based approach used to identify root causes, implement corrective actions, and prevent recurrence of quality issues. Originally developed in the automotive industry, 8D is now widely adopted across manufacturing and regulated industries to meet the corrective action requirements of ISO 9001, IATF 16949, and FDA […]

How to Use Key Risk Indicators to Manage Risks and Improve Performance
Risk is inevitable in any business, but it can also be a source of opportunity and competitive advantage. You may have tried to use traditional risk management approaches, where each business unit leader is responsible for managing risks within their area of responsibility. Discover how to mitigate risk with Key Risk Indicators. […]

ISO 19011: A Comprehensive Guide to Quality Management Auditing
Summary: ISO 19011 provides comprehensive guidelines for planning, conducting, and improving management system audits across any standard, including ISO 9001, IATF 16949, and ISO 14001. It establishes the principles of effective auditing, defines auditor competence requirements, and offers a structured approach to managing audit programs that drive continuous improvement. Following ISO 19011 guidelines helps organizations […]
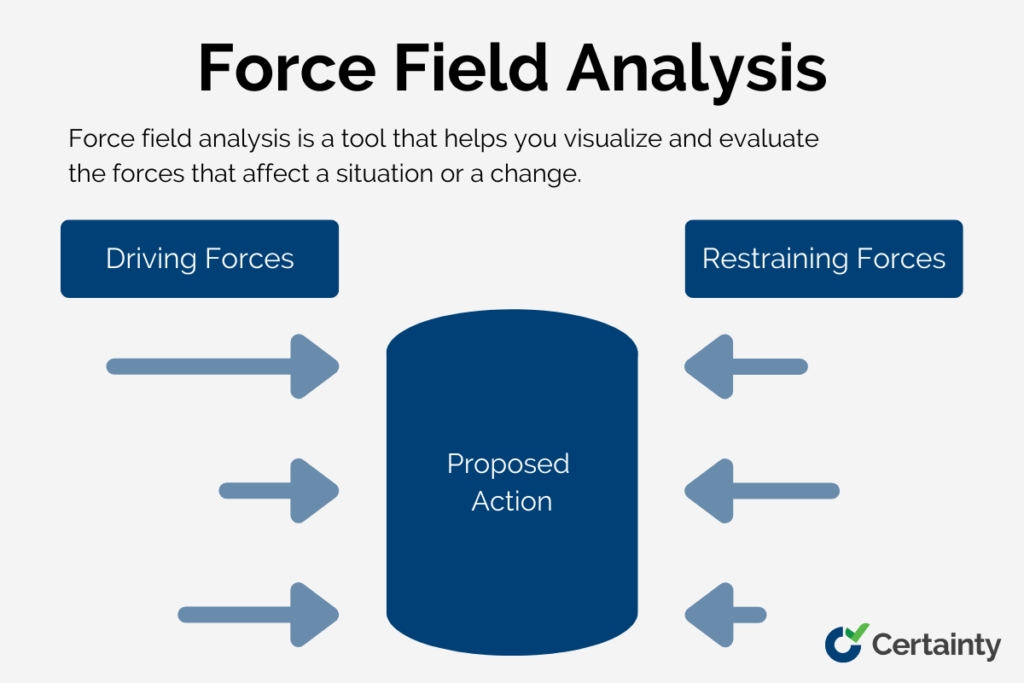
How to Use Force Field Analysis to Manage Change and Improve Performance
Summary: Force field analysis is a change management tool that identifies the driving forces supporting a desired change and the restraining forces working against it. By systematically mapping and scoring these opposing forces, quality teams can develop targeted strategies to strengthen drivers and weaken barriers, leading to more successful process improvements. This technique is particularly […]




