Summary: ISO 19011 provides comprehensive guidelines for planning, conducting, and improving management system audits across any standard, including ISO 9001, IATF 16949, and ISO 14001. It establishes the principles of effective auditing, defines auditor competence requirements, and offers a structured approach to managing audit programs that drive continuous improvement. Following ISO 19011 guidelines helps organizations […]
Quality Management
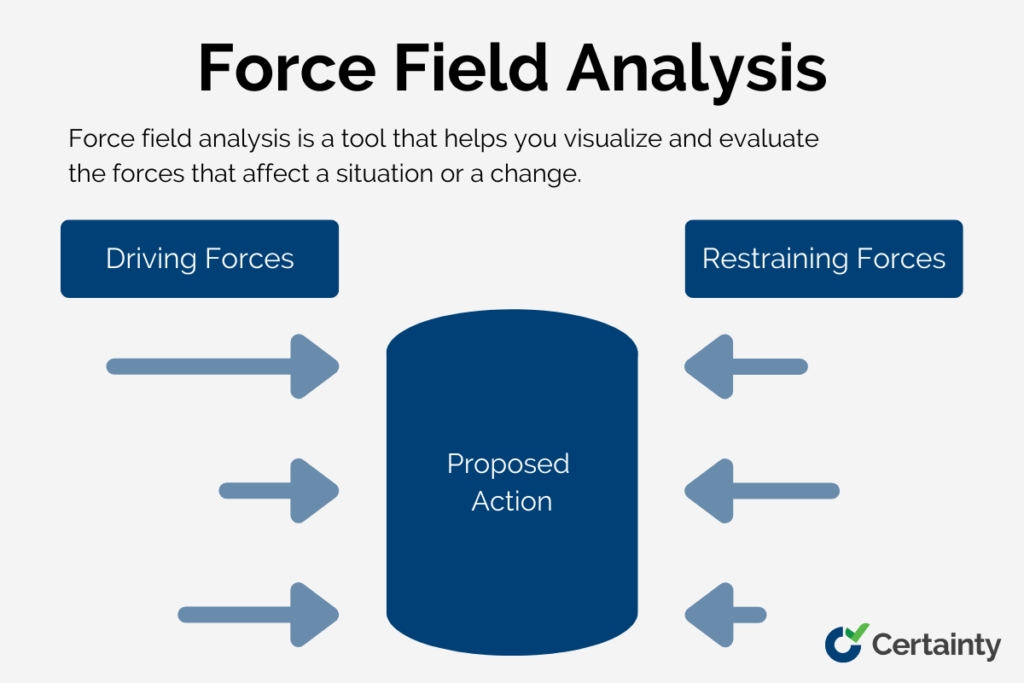
How to Use Force Field Analysis to Manage Change and Improve Performance
Summary: Force field analysis is a change management tool that identifies the driving forces supporting a desired change and the restraining forces working against it. By systematically mapping and scoring these opposing forces, quality teams can develop targeted strategies to strengthen drivers and weaken barriers, leading to more successful process improvements. This technique is particularly […]

Certainty and AI: Implementing AI in Certainty to Reduce Risk, Improve Performance, and Fuel Innovation and Growth
In this blog post, we will share the benefits of implementing AI in Certainty to enhance our product features and benefits for our customers. We will also explain some tips to best prepare your AI implementation. […]

10 Things to Consider When Selecting a Layered Process Audit Tool
Summary: Layered Process Audits (LPAs) are a proven quality management technique where audits are conducted at multiple management levels to verify that standardized processes are followed consistently. Selecting the right LPA tool is critical for reducing audit fatigue, ensuring cross-site comparability, and driving down non-conformance rates across manufacturing operations. The ideal tool should support mobile […]

Bridging the Gap: 10 Strategies for Closing Communication Gaps During Internal Audits and Inspections
Effective communication is crucial for the success of any business, and ensuring that all team members are on the same page and understand their roles and responsibilities is vital for achieving compliance and improving overall organizational performance. However, communication breakdowns can occur in any organization, and they can be particularly damaging when it comes to internal audits and inspections. […]

Internal Audits and Inspections System: A Guide to Change Management
This blog is intended to serve as a guide for those leading their organizations through the changeover process. From project planning to ongoing support, we will discuss the key steps necessary in change management. You’ll learn how to successfully implement your new system while minimizing your risk of negative consequences […]
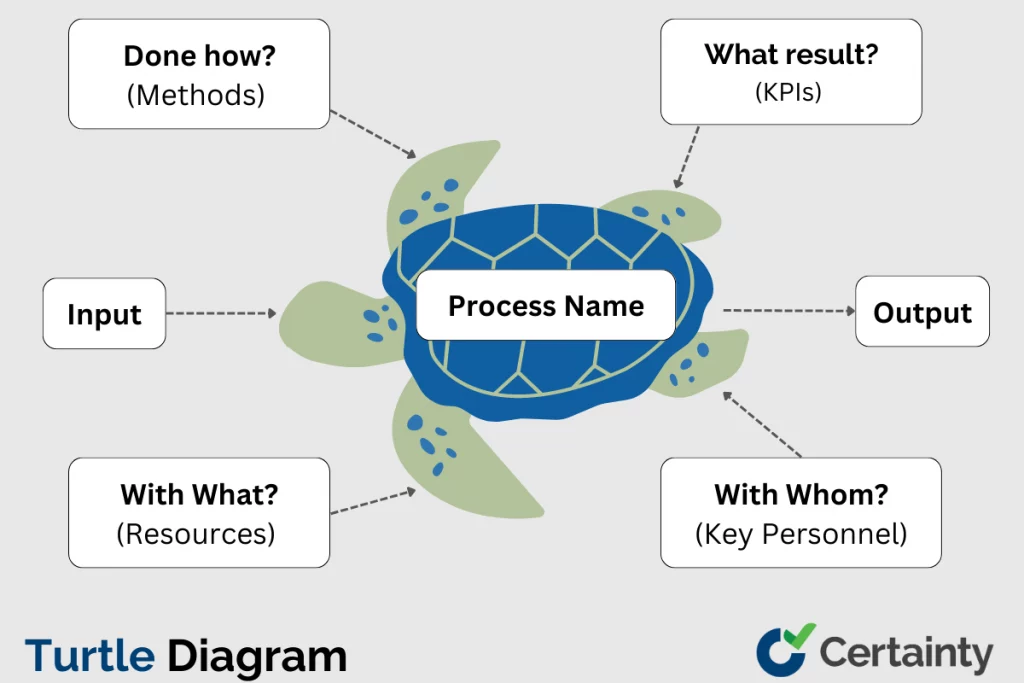
Turtle Diagram: Simplifying Your QMS Processes
Summary: A Turtle Diagram is a visual quality management tool that maps the inputs, outputs, resources, and controls of any process in a simple, intuitive format. It is widely used during ISO 9001, IATF 16949, and VDA 6.3 process audits to evaluate whether processes are adequately defined, resourced, and performing as intended. By breaking complex […]

IATF 16949: Quality Management in the Automotive Manufacturing Industry
Summary: IATF 16949 is the global quality management system standard developed specifically for the automotive industry, building on ISO 9001 with additional requirements for defect prevention, variation reduction, and supply chain management. Certification demonstrates a manufacturer's commitment to producing safe, reliable automotive components while meeting the stringent expectations of OEMs worldwide. Implementing IATF 16949 drives […]

5 Challenges When Switching From Paper-based Inspections to Digital Inspections
Table of contents Summary: Digital inspections help teams replace paper forms with faster, more consistent, and easier-to-report inspection processes. While the transition can involve change management, training, device costs, and security concerns, those challenges can be managed with the right rollout plan. Digital inspections help organizations replace slow, error-prone paper processes with faster data capture […]

ISO 9001: an Overview of the Quality Management System Standard
Summary: ISO 9001 is the internationally recognized standard for quality management systems, certified by over one million organizations across 170+ countries. It provides a framework for building consistent, customer-focused processes through a Plan-Do-Check-Act cycle of continuous improvement. Organizations that achieve ISO 9001 certification can reduce non-conformance rates, lower the cost of poor quality, and demonstrate […]

What is VDA 6.3?
Summary: VDA 6.3 is a process audit standard developed by the German Association of the Automotive Industry (VDA) that evaluates manufacturing and service processes across the automotive supply chain for quality compliance. For Quality Engineers, QA Managers, and Lead Auditors in the automotive sector, VDA 6.3 builds on ISO 9001 and IATF 16949 requirements to […]

The CAPA Process: Six Steps for Improved Quality Management
Summary: The CAPA (Corrective and Preventive Action) process provides a six-step framework for identifying non-conformances, determining root causes, implementing corrections, and preventing recurrence — making it one of the most powerful tools available for reducing cost of poor quality (COPQ). Required under ISO 9001 Clause 10.2, FDA cGMP (21 CFR Part 820), and IATF 16949, […]

What is an ISO Audit — and Why Does Your Business Need One?
Summary: An ISO audit is a systematic, independent evaluation of your organization's practices against internationally recognized standards for quality management, environmental management, or information security. For QA Managers, Quality Directors, and Lead Auditors, ISO audits validate that quality management systems are functioning effectively and drive measurable improvements in non-conformance rates, audit completion rates, and time […]

Quality Control Inspections: 5 Common Types to Boost Your QMS
Summary: Quality control inspections — including pre-production, in-line, final, and incoming inspections — verify that products meet established standards at every stage of the manufacturing process. For QA Managers and Quality Engineers tracking first pass yield (FPY), cost of poor quality (COPQ), and non-conformance rates under ISO 9001, IATF 16949, or FDA cGMP, implementing the […]

Audit Types Explained: What You Need to Know About First, Second, and Third-Party Audits
Auditing is a process of verifying and evaluating a business’s compliance, quality, and performance. However, there are different types of audits that serve different purposes and have different requirements. In this blog post, you will learn what first-party, second-party, and third-party audits are, how they differ, and why they are important for your business […]




