Summary: Quality Assurance (QA) and Quality Control (QC) are complementary but distinct disciplines within a quality management system — QA focuses on preventing defects through process design and standardization, while QC detects defects through testing and inspection of finished outputs. Together, they form a comprehensive approach to quality that drives first pass yield improvements, reduces the cost of poor quality, and ensures compliance with standards like ISO 9001 and IATF 16949. Understanding when and how to apply each discipline helps quality teams allocate resources effectively and build a culture of continuous improvement.
Table of contents

Whether in manufacturing, pharmaceuticals, or other regulated industries, delivering high-quality products is a cornerstone of business success. Quality management processes like Quality Assurance (QA) and Quality Control (QC) are pivotal in ensuring consistency, reliability, and customer satisfaction across every stage of the production lifecycle.
Yet, these terms are often misunderstood by quality teams, leading to confusion, inefficiencies, and missed opportunities for improvement. For QA Managers, Quality Engineers, and Plant Managers tasked with meeting standards like ISO 9001 or FDA cGMP, the distinction is more than semantic — it directly impacts audit outcomes, non-conformance rates, and cost of poor quality (COPQ).
Understanding the distinction between QA and QC is the foundation of better decision-making and operational efficiency for Quality Supervisors, Process Improvement Leads, and VP-level quality leaders driving compliance across multiple sites.
This article will clarify the differences between QA and QC, explore how they work together, and offer actionable strategies for implementing both in your organization to improve first pass yield (FPY) and reduce time to resolution on corrective actions.
Why Quality Matters in Today’s Enterprises
Superior quality can be the difference maker that makes the final product stand out from your competitors in a highly competitive field — and it is a metric that QA Managers and Plant Managers are measured against every day.
Why is that? High-quality products build customer trust, foster loyalty, and enhance brand reputation. Conversely, lapses in quality can lead to devastating consequences, such as regulatory fines, product recalls, or loss of market share. For organizations tracking quality KPIs like non-conformance rates, audit completion rates, and COPQ, any gap in the quality system is a gap in the bottom line.
Take the pharmaceutical manufacturing process as an example. Regulatory bodies like the FDA and EMA enforce stringent quality requirements — including FDA cGMP compliance — to ensure public safety. Failing to meet these standards can result in costly recalls or legal actions. Quality Engineers and Lead Auditors working under these frameworks know that audit readiness is not optional; it is a continuous requirement.
Similarly, in the automotive industry, a single manufacturing defect can cascade into massive recalls and reputational damage. Organizations operating under IATF 16949 and VDA 6.3 standards must maintain rigorous process audits to prevent systemic failures. For instance, Takata’s recall for faulty airbags in millions of vehicles led to one of the most expensive recalls in history, costing the manufacturer billions of dollars and ultimately filing for bankruptcy. These cases underscore the critical importance of robust quality systems.
For industries like food and beverage, quality failures can have life-threatening consequences. Contaminated products not only harm consumers but also result in eroded trust and significant financial losses. Companies that excel in quality management — maintaining HACCP compliance and tracking FPY across production lines — consistently exceed customer expectations while maintaining compliance with safety regulations.
Defining Quality Assurance (QA) and Quality Control (QC)
What is Quality Assurance?
Quality Assurance is a proactive and systematic approach to quality management. The primary goal of quality assurance is to design and implement processes that prevent defects from occurring in the first place. Unlike QC, which focuses on the end product, QA emphasizes process improvement and consistency throughout the production lifecycle — a core principle embedded in ISO 9001’s risk-based thinking and the Plan-Do-Check-Act (PDCA) cycle.
For example, in food manufacturing, QA might involve developing comprehensive HACCP (Hazard Analysis and Critical Control Points) plans to identify and mitigate risks at every stage of production. In the automotive sector, QA could mean conducting VDA 6.3 process audits on supplier facilities. In electronics manufacturing, QA includes setting design guidelines to ensure components meet performance and reliability standards before production begins.
Stuck with managing HACCP compliance? Access Certainty Software’s free-to-download HACCP Food Safety Checklist today.

30+ Audit and inspection checklists free for download.
What is Quality Control?
Quality Control, on the other hand, is reactive. It involves inspecting and testing products to identify and correct defects after they occur. QC serves as the final checkpoint, ensuring that only products meeting the required specifications reach the customer. For Quality Supervisors and QC inspectors, this means managing non-conformance reports, tracking defect rates, and ensuring time to resolution stays within acceptable limits.
For instance, in automotive manufacturing, QC teams might inspect vehicles for paint defects, engine performance issues, or electrical malfunctions. In pharmaceutical environments governed by FDA cGMP, QC includes testing each batch for potency, purity, and sterility. These inspections are crucial for identifying problems that could lead to recalls or customer dissatisfaction.
While QC is essential for maintaining quality standards, it works best when paired with strong QA practices. Together, they form a comprehensive system for managing quality across all stages of product development — and when supported by digital tools, they eliminate the inefficiencies of paper-based audits and manual corrective action tracking.
Key Differences Between Quality Assurance and Quality Control
Understanding the differences between QA and QC is essential for optimizing your quality management strategy. Here’s a side-by-side comparison that QA Managers and Quality Engineers can use to align team responsibilities:
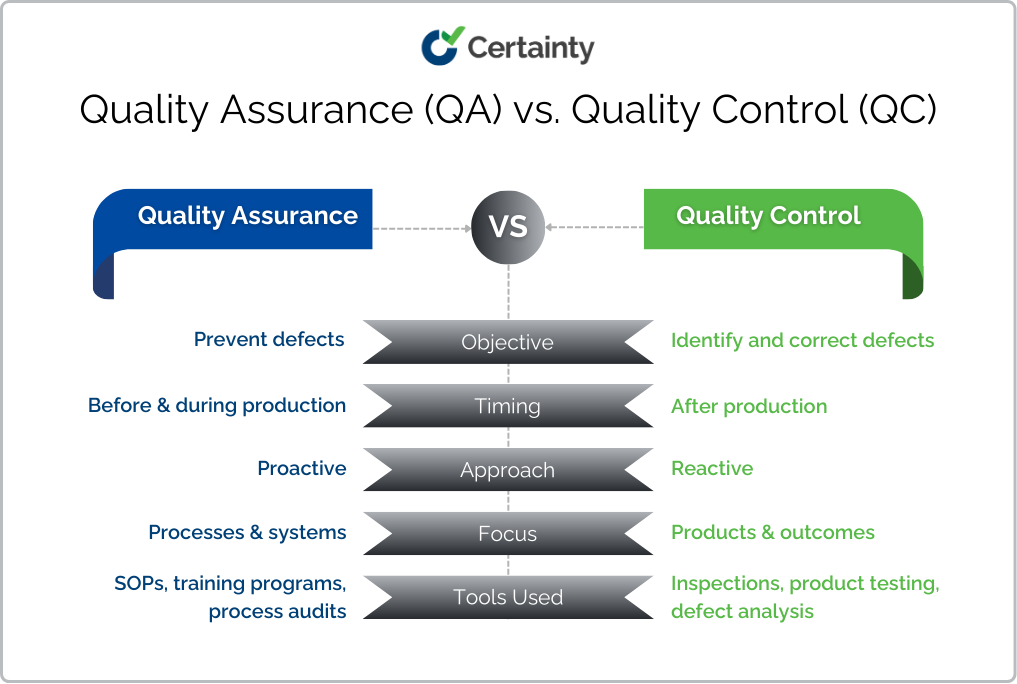
While QA lays the foundation for quality through prevention, QC provides the safety net by catching errors before they reach the customer. Both are critical for achieving consistent and reliable outcomes — and for driving measurable improvements in KPIs like first pass yield and audit completion rates.
How QA and QC Work Together
QA and QC do serve different purposes — but they are most effective when used in tandem. A strong quality management system integrates the strengths of both approaches, creating a feedback loop that drives continuous improvement and aligns with the corrective and preventive action (CAPA) requirements found in ISO 9001 and FDA cGMP frameworks.
Consider an electronics manufacturing company. QA activities might involve designing a production process that minimizes stress on sensitive components, while the QC process involves testing finished devices to ensure durability under various conditions. The insights gained from QC — such as recurring non-conformance patterns or shifts in defect rates — can then inform QA processes, leading to further refinements and fewer defects over time. For Process Improvement Leads, this feedback loop is where the real gains in FPY and COPQ reduction happen.
Benefits of Effective QA and QC Strategies
Implementing robust QA and QC strategies offers numerous benefits, including cost savings, improved compliance, and enhanced customer satisfaction — outcomes that directly impact the metrics QA Managers and VP Quality Assurance leaders report on.
For example, a food manufacturer combining QA protocols (like HACCP plans) with rigorous QC methodologies (such as microbial testing) can prevent contamination while ensuring regulatory compliance. This dual approach reduces the risk of recalls, strengthens brand reputation, and fosters customer loyalty. Organizations tracking COPQ often find that every dollar invested in prevention through QA yields significant returns by reducing rework, scrap, and warranty claims.
In addition to these tangible benefits, effective quality assurance and quality control activities also improve operational efficiency. By reducing defects, deviations, and rework, companies will save time, lower their production costs, and increase their overall return on investment. Quality teams that move away from paper-based audits and adopt digital inspection tools gain real-time visibility into audit completion rates, non-conformance trends, and corrective action status across all sites.
Challenges in Implementing QA and QC (And How to Overcome Them)
Despite their importance, implementing QA and QC systems can be challenging. Common pitfalls include over-reliance on QC while neglecting QA, lack of employee training, resistance to change, and audit fatigue — where teams conducting frequent inspections across multiple sites lose consistency and thoroughness due to repetitive manual processes.
Many organizations also struggle with paper-based audit workflows that make cross-site comparability nearly impossible. When QA Managers and Lead Auditors cannot easily compare findings across facilities, non-conformances slip through the cracks and corrective actions stall. Manual corrective action processes — where follow-ups are tracked in spreadsheets or email — further delay time to resolution and increase the risk of recurring issues.
One way to overcome these challenges is to leverage technology. Tools like Certainty Software can streamline inspections, audits, and reporting, making it easier to not only employ corrective actions but also maintain consistency across teams and locations. Digitizing your audit workflows eliminates the cross-site comparability gap and gives Quality Engineers and Plant Managers real-time dashboards for tracking non-conformance rates and audit completion. Additionally, fostering a culture of quality — where employees at all levels understand their role in maintaining standards — can significantly enhance the effectiveness of QA and QC systems.
Real-World Examples of QA and QC
Let’s look at how QA and QC play out in different industries:
- Pharmaceutical Manufacturing: QA involves creating protocols aligned with FDA cGMP to maintain cleanroom conditions and validate processes, while QC includes testing each batch of products for potency and purity — tracking non-conformance rates to ensure every lot meets release specifications.
- Automotive Manufacturing: QA focuses on supplier audits under IATF 16949 and VDA 6.3 process design reviews, while QC inspects finished vehicles for defects like engine faults or paint imperfections. Quality Engineers use FPY data from QC to refine upstream QA processes.
- Food and Beverage Manufacturing: QA designs HACCP plans and conducts internal audits to maintain compliance, and QC tests finished products for microbial contamination — with audit completion rates tracked across every production facility.
- Electronics Manufacturing: QA sets design standards and conducts process validation per ISO 9001, while QC conducts stress tests on devices to identify hardware failures and measures COPQ to quantify the cost impact of defects.
How Certainty Software Helps You Manage QA and QC
Quality Assurance and Quality Control are complementary components of an effective quality management system. As you grow to understand their unique roles and integrate them effectively, your organization can achieve higher efficiency, stronger compliance with standards like ISO 9001, IATF 16949, and FDA cGMP, and greater customer satisfaction.
To optimize your quality management processes, explore how Certainty Software’s tools can streamline QA and QC activities — from digitizing paper-based audits and automating corrective action workflows to providing real-time dashboards for tracking audit completion rates, non-conformance trends, and COPQ across all your sites. Certainty ensures your organization stays ahead in today’s competitive market and meets global regulatory requirements.
Schedule your free call today with our team to learn more about how Certainty can support your quality management processes.
Frequently Asked Questions (FAQs)
What is the main difference between QA and QC in quality management?
Quality Assurance (QA) is a proactive, process-oriented approach focused on preventing defects by designing and improving production processes. Quality Control (QC) is a reactive, product-oriented approach focused on identifying and correcting defects through inspection and testing. QA asks “are we building the product the right way?” while QC asks “does the finished product meet our specifications?” Both are essential components of a complete quality management system under frameworks like ISO 9001.
How do QA and QC relate to ISO 9001 and other quality standards?
ISO 9001 requires organizations to implement both preventive (QA) and detective (QC) quality measures as part of their quality management system. QA aligns with ISO 9001’s emphasis on risk-based thinking, process approach, and the Plan-Do-Check-Act cycle. QC supports the monitoring, measurement, and evaluation requirements. Industry-specific standards like IATF 16949 for automotive, FDA cGMP for pharmaceuticals, and HACCP for food safety build on these principles with additional QA and QC requirements tailored to each sector.
What KPIs should QA Managers track to measure quality performance?
Key quality KPIs include first pass yield (FPY), which measures the percentage of products manufactured correctly without rework; non-conformance rates, which track defects or deviations from specifications; cost of poor quality (COPQ), which quantifies the financial impact of defects, scrap, and rework; audit completion rates, which indicate how consistently scheduled audits are being executed; and time to resolution, which measures how quickly corrective actions are closed out after a non-conformance is identified.
How can organizations overcome challenges with paper-based quality audits?
Paper-based audits create significant challenges for quality teams, including difficulty with cross-site comparability, slow corrective action follow-up, and limited real-time visibility into quality performance. Organizations can overcome these issues by digitizing their audit and inspection workflows with quality management software like Certainty. Digital tools enable standardized checklists, automated corrective action assignments, real-time dashboards, and centralized data — helping QA Managers and Lead Auditors reduce audit fatigue and ensure consistency across multiple facilities.
Can QA and QC work together to reduce cost of poor quality (COPQ)?
Yes, QA and QC form a powerful feedback loop for reducing COPQ. QC activities — such as inspections and product testing — identify defects and generate data on non-conformance patterns. QA teams then use this data to refine processes, update standard operating procedures, and implement preventive measures that address root causes. Over time, this continuous improvement cycle reduces rework, scrap, warranty claims, and recalls, driving measurable reductions in COPQ and improving overall first pass yield.
You might also be interested in:

Spend Time On Prevention
Not Paperwork
Watch our overview video to see how your organization can benefit from Certainty.
Watch Video



