Summary: An ISO 9001 audit systematically evaluates your quality management system against international standards, verifying QMS alignment, identifying non-conformances, and developing corrective actions for continual improvement. Organizations that implement ISO 9001 report up to 18% increases in client satisfaction and 15% cost reductions — making audit preparation a high-impact activity for QA Managers, Quality Engineers, and Lead Auditors tracking audit completion rates and cost of poor quality.
According to one study, businesses that implemented the ISO 9000-based quality standards found an 18% increase in client satisfaction and a 15% cost reduction. For Quality Managers, Plant Managers, and Process Improvement Leads, these gains translate directly into lower Cost of Poor Quality (COPQ) and higher first-pass yield (FPY). ISO 9001 is an internationally recognized Quality Management System (QMS) criteria program that guides businesses of any size to maintain or improve their quality management system and processes. Whether your organization also operates under IATF 16949, VDA 6.3, FDA cGMP, or HACCP requirements, ISO 9001 serves as the foundational audit framework. The ISO approach and ISO 9001 audit is based on the Deming Cycle of continuous improvement – plan, do, check, act.
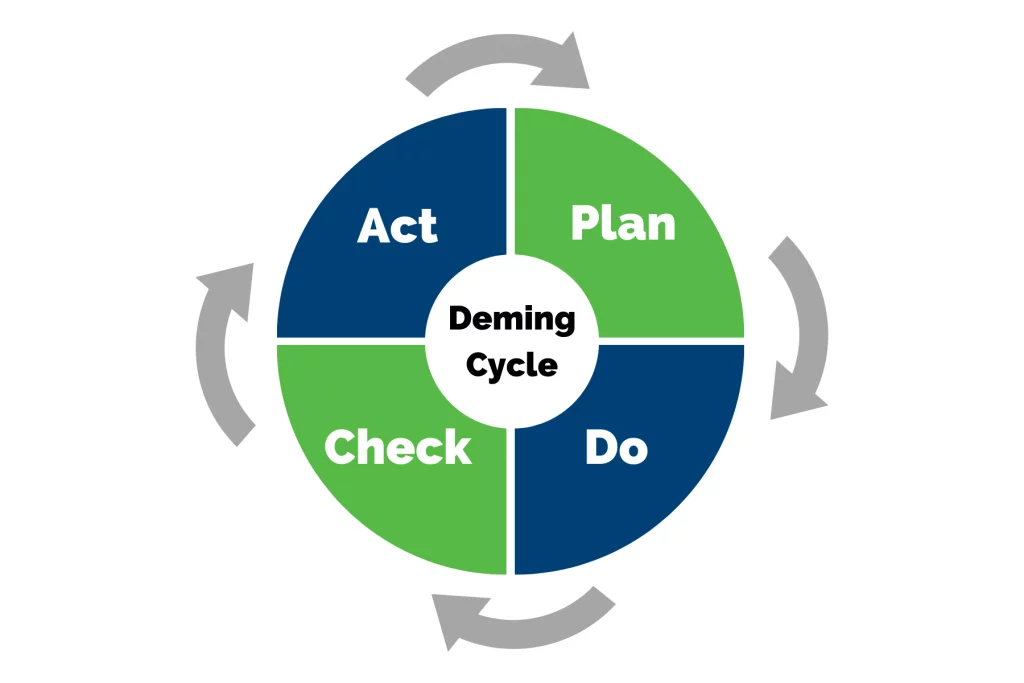
To either become certified or to maintain ISO standards once certified, auditing both externally and internally is a necessary action. For QA Directors and Quality Engineers tracking audit completion rates and non-conformance trends, the ISO 9001 audit aims to address three key areas of your Quality Management System:
- Verify your QMS aligns with the ISO 9001 audit standards.
- Identify areas of concern within your quality system.
- Develop corrective actions and opportunities for improvement.
What are the 3 Types of ISO 9001 Audits?
An ISO 9001 audit can be divided into either general internal or external auditing. Internal auditing is conducted within the business and holds accountability to meet the standards created by ISO. External audits are scheduled and completed by auditors external to the business. External auditors offer precision knowledge of ISO standards and opportunities for new perspectives on your business’s Quality Management Systems.
There are three options that businesses have when choosing how to conduct their ISO 9001 audit:
Self-auditing does not necessarily mean an audit is completed by management or employees. Rather, it also includes reflecting on the feedback that a business’s customers have provided, or a surveillance audit. For Quality Supervisors managing multiple facilities, self-audits also present an opportunity to benchmark cross-site comparability and identify systemic gaps before an external auditor does.
On-site audits are pre-scheduled and typically take at least a full business day to conduct. Auditing completion time varies on factors such as business size, QMS complexity within the business, and other factors. The external ISO 9001 audit frequency varies but is typically conducted on an annual basis. Digital audit tools can dramatically reduce on-site audit duration by enabling real-time data capture and automated checklist completion, directly improving your audit completion rates.
Remote audits can be completed through web meetings, phone calls, and electronic document transfers. With modern digital audit platforms, remote audits have become far more effective, enabling Lead Auditors to review evidence, verify corrective actions, and maintain audit trails electronically – reducing audit fatigue for both auditors and auditees.
What is the Internal ISO 9001 Audit Process?
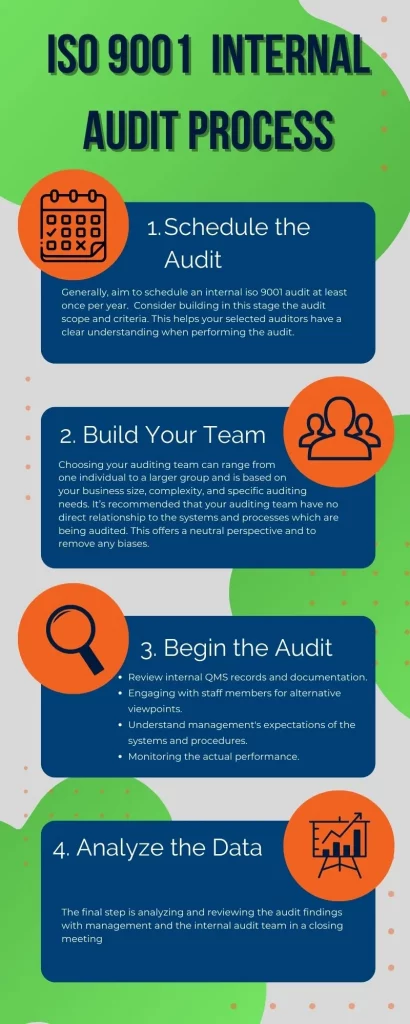
The ISO 9001 internal audit process is a self-check opportunity to ensure your QMS and processes meet the ISO standards. For VP Quality Assurance leaders and QA Managers overseeing multi-site operations, a structured internal audit process is critical to reducing non-conformance rates and driving down COPQ. For a more effective internal ISO 9001 audit, it is recommended to follow these four steps:
1. Schedule the Audit
Generally, aim to schedule an internal ISO 9001 audit at least once per year. Of course, this varies based on the complexity of your quality management systems and may require more frequent internal auditing. Organizations that also maintain IATF 16949 or FDA cGMP certifications often require quarterly or even monthly internal audit cycles. In addition to building the audit schedule, consider building the audit scope and criteria. This helps your selected auditors have a clear understanding when performing the audit. A digital audit scheduling tool ensures nothing slips through the cracks and provides cross-site visibility into audit completion rates.
2. Construct the Audit Team
Choosing your auditing team can range from one individual to a larger group and is based on your business size, complexity, and specific auditing needs. It’s recommended that your auditing team have no direct relationship to the systems and processes that are being audited. This offers a neutral perspective and removes any biases.
Selecting a neutral auditing team with no direct involvement in your QMS also creates a challenge for the auditing process. The lack of quality system experience by the internal auditor(s) could result in key audit findings being missed and a failure to meet the requirements of ISO 9001 unless clear auditing directions are given.
To avoid setbacks from this challenge, we recommend using a quality assurance checklist that covers all necessary auditing components and reduces missed observations by your auditing team. Standardized digital checklists eliminate the inconsistencies of paper-based audits and ensure cross-site comparability across all your facilities. Additionally, we recommend conducting a series of internal auditor training to familiarize your internal auditors with the procedures and tools required for a well-performed audit.
3. Begin the ISO 9001 Audit
Now that you’ve assembled your ISO internal audit team, and the auditors have been briefed thoroughly at your opening meeting on the auditing process and objectives, the actual auditing process may begin. For the ISO internal audit to be a success, we recommend ensuring the following 4 tasks are audited:
- Observing internal records and documentation related to QMS.
- Engaging with staff members for alternative viewpoints on performance.
- Developing an understanding of management expectations of the systems and procedures.
- Monitoring the actual performance of the systems and procedures.
Having to examine many different systems and procedures associated with the internal ISO 9001 audit can create hurdles for your auditing team. More specifically, if they aren’t given the supportive audit tools to enter observations in real time, delays in audit completion are more likely to occur. Replacing paper-based audit forms with digital tools enables real-time data capture, automatic non-conformance flagging, and instant corrective action assignment – reducing your time to resolution and improving overall audit accuracy. Also, tools for streamlined data prevent having to recall and input observations at a later time, improving your audit result accuracy, and ultimately meeting the standards set by ISO.
4. Analyze the Data
The final step is analyzing and reviewing the audit findings with management and the internal audit team in a closing meeting.
A common barrier to success at this stage is being unable to organize the data into clear and actionable reports quickly. Quality Engineers and Process Improvement Leads need dashboards that surface key metrics – non-conformance rates, COPQ trends, and time to resolution – without manual data manipulation. Centralizing the audit data to output meaningful information for management review supports a continuous improvement culture in addition to conforming to the QMS standards set out by ISO. We recommend finding a software solution that is capable of centralizing your internal ISO 9001 audit data in real-time, and into simple to-analyze reports – making your next ISO internal audit a huge success.
Tips to Prepare for an External ISO 9001 Audit
Perform Internal Audits
Internally verify that your QMS is meeting the ISO certification standards by performing internal audits and check-ins. Typically, internal audit programs are the first step to recognizing when quality standard processes and/or systems are failing to meet ISO standards. For organizations also maintaining IATF 16949 or VDA 6.3 compliance, internal audits serve double duty by satisfying multiple framework requirements simultaneously.

30+ Audit and inspection checklists free for download.
Verify Documentation
As the auditee, any relevant documentation involved in your quality management system should be approved and up-to-date. Suggested documents to verify include:
- Flowcharts
- Quality objectives
- Instruction manuals
- Policies
- Records
- Corrective action logs and non-conformance reports
- Previous audit findings and evidence of closure
Train Employees
Employees should be aware and trained for the ISO 9001 audit process and what they can expect during the audit procedure – such as employee interviews. Additionally, training on organizational quality procedures and objectives should be an ongoing process regardless of upcoming ISO 9001 external audits.
Maintain a Culture of Continuous Improvement
When an organization embodies a continuous improvement culture, it translates into better performance for both the ISO certification audit and the ISO standard verification audit. This culture welcomes employee engagement and input into quality systems and provides new perspectives that may have been unforeseen by senior management. For Quality Managers tracking KPIs such as FPY, COPQ, and non-conformance rates, a continuous improvement culture ensures these metrics trend in the right direction – well before an external auditor arrives.
Frequently Asked Questions (FAQs)
How often should an ISO 9001 internal audit be conducted?
Most organizations conduct internal ISO 9001 audits at least once per year, but the ideal frequency depends on your QMS complexity, the number of sites you operate, and whether you hold additional certifications such as IATF 16949 or FDA cGMP. High-risk processes or areas with recurring non-conformances may warrant quarterly audits. Tracking audit completion rates across all facilities helps QA Managers ensure no location falls behind schedule.
What is the difference between an ISO 9001 internal audit and an external audit?
An internal audit is performed by your own team (or contracted auditors working on your behalf) to verify that your QMS conforms to ISO 9001 requirements before an external review. An external audit is conducted by an accredited certification body to formally assess compliance and grant or maintain certification. Internal audits give Quality Supervisors and Process Improvement Leads the chance to identify and resolve non-conformances, reduce COPQ, and improve time to resolution before external auditors evaluate your systems.
How can digital audit tools improve ISO 9001 compliance?
Digital audit tools replace paper-based audit processes with real-time data capture, automated corrective action workflows, and centralized reporting dashboards. This eliminates the inconsistencies of manual data entry, reduces audit fatigue for your team, and enables cross-site comparability so Plant Managers and VP Quality Assurance leaders can benchmark performance across every facility. The result is faster audit completion, lower non-conformance rates, and measurable reductions in COPQ.
What are common non-conformances found during an ISO 9001 audit?
Common non-conformances include inadequate document control, incomplete corrective action records, lack of management review evidence, insufficient employee training documentation, and failure to monitor key quality KPIs such as FPY and non-conformance rates. Many of these findings stem from reliance on paper-based systems that make it difficult to maintain audit trails. A digital quality management platform helps Quality Engineers proactively identify and close these gaps before they become formal findings.
How does ISO 9001 relate to other quality standards like IATF 16949 and FDA cGMP?
ISO 9001 provides the foundational QMS framework that many industry-specific standards build upon. IATF 16949 extends ISO 9001 for the automotive sector, VDA 6.3 adds process audit requirements for automotive suppliers, and FDA cGMP applies ISO-aligned principles to pharmaceutical and medical device manufacturing. Organizations holding multiple certifications benefit from a unified audit platform that maps shared clauses across frameworks, reducing duplicate effort and audit fatigue while maintaining compliance across all applicable standards.
You may also be interested in:
What Are Quality Assurance Inspections And Why They Matter
Improving Operational Quality: The Role Of Qualitative Risk Assessment
Solutions for Improving Quality Assurance & Quality Control

Spend Time On Prevention
Not Paperwork
Watch our overview video to see how your organization can benefit from Certainty.
Watch Video



