
A Layered Process Audit (LPA) is a structured quality auditing method in which multiple organizational levels — from frontline operators to senior executives — each conduct frequent, standardized audits of critical manufacturing or service processes using pre-defined checklists. Unlike product audits, which inspect finished output, LPAs verify process inputs, machine settings, operator behaviors, and procedural compliance at the point of work, catching deviations before they produce defects. The “layered” structure means each level provides a different frequency and scope of oversight: daily at the operator layer, weekly at the supervisor layer, and monthly or quarterly at management and executive layers.
LPAs are most commonly associated with automotive manufacturing — where the AIAG CQI-8 Layered Process Audit Guideline is the canonical reference — but the methodology is used across aerospace, medical devices, food and beverage, and any industry where process discipline determines product quality.
LPA at a glance
| Who does them | All management layers — operators, supervisors, managers, executives |
| How often | Daily (Layer 1), weekly (Layer 2), monthly (Layer 3), quarterly (Layer 4) |
| What’s audited | Process inputs, methods, settings, operator behaviors — not finished product |
| Standard reference | AIAG CQI-8 (2nd Edition) |
| Typical outcomes | Lower defect rates, reduced Cost of Poor Quality, stronger quality culture |
| Required by | OEM Customer Specific Requirements (e.g., GM, Stellantis), not IATF 16949 directly |
Table of contents
- Why LPAs matter: the business case
- The 4 layers of an LPA
- LPA vs. Gemba walk vs. 5S audit vs. internal audit
- CQI-8 2nd Edition: the definitive LPA standard
- Are LPAs required by IATF 16949?
- How to implement an LPA program (8 steps)
- Writing effective LPA checklist questions
- Preventing pencil whipping and audit fatigue
- LPA KPIs and benchmarks
- LPAs beyond automotive
- LPA software: what to look for
- Frequently asked questions
- Related resources

Why Layered Process Audits matter
The business case in one paragraph
Most quality failures are not caused by unknown risks — they are caused by known risks that are not consistently controlled. A machine setting drifts. An operator skips a verification step under time pressure. A control plan is updated but the work instruction is not. LPAs are the mechanism that catches these everyday deviations before they produce defects, warranty claims, recalls, or customer escapes.
The cost of poor quality
The Automotive Industry Action Group (AIAG) estimates that the Cost of Poor Quality (CoPQ) — scrap, rework, warranty, inspection, field failures — typically runs 15–20% of sales revenue in manufacturing organizations. LPAs directly attack CoPQ by shifting defect prevention earlier in the process, where corrective cost is lowest. The 1-10-100 rule of quality illustrates the math: a defect caught at the operator layer costs $1 to fix; caught at end-of-line inspection, $10; caught by the customer, $100+.
Four outcomes mature LPA programs consistently produce
- Defect rate reduction. Published case studies from automotive suppliers commonly cite 30–50% defect rate improvements within 12 months of a disciplined LPA rollout.
- First-pass yield improvement. Because deviations are caught and corrected in-process, fewer units require rework.
- Management engagement in quality. LPAs require managers and executives to physically visit the floor — a behavior change that compounds cultural benefits beyond the audit itself.
- Audit trail for compliance. LPA records demonstrate evidence of internal process audits required by ISO 9001:2015 Clause 9.2, IATF 16949:2016, and many regulatory frameworks.
For a deeper treatment, see our key benefits of layered process audits article.
The 4 layers of an LPA
The “layers” are the defining feature of an LPA program — each organizational level conducts its own audits at its own frequency, with its own scope. Together, they create redundant, cascading oversight that no single audit event could provide.

Layer 1 — Operators, Team Leaders, and Shift Supervisors
Frequency: Daily (often per-shift)
Checklist length: 10–15 binary (yes/no) questions
Scope: Immediate process compliance — machine settings, tooling, work instructions, PPE, 5S condition, startup/changeover verification
Layer 1 auditors are closest to the process. Their proximity enables the fastest detection of deviations. These audits are short by design — long checklists at Layer 1 invite “pencil whipping” (see Preventing pencil whipping below).
Layer 2 — Supervisors, Engineers, and Cross-functional Staff
Frequency: Weekly or bi-weekly
Checklist length: 15–25 questions
Scope: Verify Layer 1 findings have been acted upon; broader process drift checks; cross-workstation trends
Layer 2 brings cross-functional perspective. An engineer auditing a cell may spot that a process step is impractical as specified — a finding an operator would accept as “just how it is.” The layer’s purpose is to elevate issues that Layer 1 cannot resolve alone.
Layer 3 — Plant Managers and Department Directors
Frequency: Monthly
Checklist length: 20–30 questions, often including systemic items
Scope: Systemic trends, multi-department patterns, program-level effectiveness, resource adequacy
Layer 3 audits are less about catching individual deviations and more about verifying that the LPA program itself is producing improvements. Are Layer 1 and 2 findings being closed? Are repeat non-conformances decreasing? Does the checklist need updates?
Layer 4 — Senior Executives and the C-Suite
Frequency: Quarterly (some organizations conduct them monthly at a rotating site)
Scope: Strategic alignment — does the LPA program support business goals? Are we allocating enough resource? What does the culture signal to the workforce?
Executive participation is the element that distinguishes mature LPA programs. The DuPont Bradley Curve — a safety/quality culture maturity model — shows that organizations without visible leadership engagement remain in “dependent” culture, where compliance is extrinsic. Executive LPA participation is the most reliable lever to move from dependent to interdependent culture, where quality ownership is distributed across the organization.

LPA vs. Gemba walk vs. 5S audit vs. internal audit
Quality and continuous-improvement practitioners often ask where LPAs fit alongside other shop-floor observation practices. They are complementary, not interchangeable.

| Attribute | Layered Process Audit | Gemba Walk | 5S Audit | Internal (QMS) Audit |
|---|---|---|---|---|
| Primary purpose | Verify process compliance | Observe work as actually done | Verify workplace organization | Verify QMS conformance |
| Structure | Structured checklist | Observational, open-ended | Structured 5S criteria | Formal audit protocol |
| Frequency | Daily to quarterly (by layer) | Weekly to monthly | Weekly to monthly | Annual or semi-annual |
| Conducted by | All management layers | Managers & leaders | Supervisors, team leaders | Trained QMS auditors |
| Output | Non-conformances → CAPA | Observations & dialogue | Score + improvement list | Formal audit report |
| Standard reference | AIAG CQI-8 | Toyota Production System | Lean/TPS / 6S Lean | ISO 9001 Clause 9.2, ISO 19011 |
| Documentation | Detailed records required | Light notes | Scorecard | Full documentation set |
When to use each
- Use LPAs when you need structured, frequent, multi-level verification of critical process parameters.
- Use Gemba walks when leadership needs to understand how work actually happens — to identify waste, improvement opportunities, and disconnects between documented and actual practice.
- Use 5S audits when the focus is workplace organization and visual management. (Note: 5S items are commonly included within Layer 1 LPA checklists rather than audited separately.)
- Use internal QMS audits for periodic, system-level verification against ISO 9001, IATF 16949, AS9100, ISO 13485, or another standard.
A mature program uses all four — each serves a different purpose. Gemba walks and LPAs pair particularly well: Gemba walks surface issues that inform LPA checklist updates; LPAs provide the frequency and accountability that Gemba walks lack.
CQI-8 2nd Edition: the definitive LPA standard
CQI-8 is the Layered Process Audit Guideline published by the AIAG. It is the canonical reference for designing, implementing, and auditing LPA programs. CQI-8 originated in 2005 as a compilation of LPA best practices from North American automotive OEMs; the 2nd edition, released in 2014 and revised since, significantly expanded the guideline’s scope and rigor.
What CQI-8 actually contains
The CQI-8 2nd Edition document includes:
- LPA methodology and principles — definitions, objectives, differentiation from product and system audits.
- Roles and responsibilities — including the LPA Planning Team and an accompanying RASIC (Responsible, Approve, Support, Inform, Consult) chart that expands LPA ownership beyond the Quality function to Engineering, Purchasing, Program Management, and Production.
- The LPA Process Owner role — a named individual accountable for program effectiveness, distinct from the auditors themselves.
- Checksheet design guidance — detailed instructions on writing effective questions (see Writing effective LPA checklist questions below).
- KPI integration — specific guidance on measuring LPA effectiveness through leading and lagging indicators (completion rate, finding rate, closure cycle time, repeat issue rate) rather than just counting audits completed.
- Corrective action linkage — how LPA findings should flow into the organization’s CAPA system.
- Program maintenance — how to review, refresh, and expand checklists over time.
- Non-manufacturing applications — explicit guidance on applying LPAs in engineering, purchasing, service, and administrative processes, not just production.
Is CQI-8 a mandatory standard?
No — CQI-8 is a guideline, not a mandated standard. Unlike IATF 16949:2016, which is a certification standard with a formal audit body, CQI-8 is a reference document organizations adopt voluntarily or because an OEM customer requires it. The practical effect is that most automotive suppliers follow CQI-8 because OEM Customer Specific Requirements (see next section) reference it directly.
CQI-8 is available for purchase (PDF or hardcover) from the AIAG store. Training courses are also offered by AIAG and third-party trainers, typically as 1–2 day sessions for LPA Planning Team members.
Are LPAs required by IATF 16949?
Short answer: Not directly by IATF 16949:2016 itself — but yes, via the OEM Customer Specific Requirements (CSRs) that sit on top of it. Any automotive supplier to General Motors, Stellantis (FCA), Ford, or several other OEMs is required to run LPAs as a condition of doing business.
What IATF 16949 actually requires
IATF 16949:2016 Clause 9.2.2.3 (“Manufacturing process audit”) requires suppliers to audit each manufacturing process to verify its effectiveness. It does not prescribe the form that audit must take. An organization can comply with 9.2.2.3 using a non-LPA audit methodology — but in practice, most automotive suppliers use LPAs because OEM CSRs require them, and because LPAs are an efficient way to produce the daily audit evidence IATF surveillance auditors expect.
OEM Customer Specific Requirements that mandate LPAs
| OEM | CSR Clause / Reference | Requirement Summary |
|---|---|---|
| General Motors | GM CSR for IATF 16949, §9.2.2.3 | Requires LPAs conducted in accordance with CQI-8; multi-layer structure; monthly effectiveness review |
| Stellantis (FCA US / Chrysler) | Stellantis CSR §9.2.2.3 | Requires LPAs per CQI-8; documented program; Layer 3/4 participation |
| Ford Motor Company | Ford CSR for IATF 16949 (June 2025); Q1 Manufacturing Site Assessment | No explicit LPA or CQI-8 mandate in the published CSR; §9.2.2.3 requires Ford Global Manufacturing Process Standards tracked via the Special Process Tracker, Supplier Manufacturing Health Charts, and CQI-9 for heat treat. Any LPA-style obligations flow from the separate Q1 Manufacturing Site Assessment, not the CSR itself |
| Honda, Toyota, Nissan | Proprietary supplier quality manuals (not published on IATF Global Oversight) | None of the three publish an IATF 16949 CSR on IATF Global Oversight, so no CQI-8/LPA mandate is publicly verifiable. Process audits are governed instead by Honda’s SQM/G-HQS (QAV-1 audits), Toyota’s Supplier Quality Assurance Manual (rooted in Toyota Production System disciplines), and Nissan’s ANPQP/NPQP frameworks — distributed to suppliers under contract |
Action for suppliers: Check the current CSR version for each OEM customer. CSRs are updated periodically; relying on an outdated version creates certification risk. The IATF Global Oversight site maintains links to current OEM CSRs.
LPAs and ISO 9001
For suppliers outside automotive, ISO 9001:2015 Clause 9.2 requires internal audits at planned intervals. LPAs are one way to satisfy or complement this requirement. Additionally, ISO 19011:2018 (Guidelines for auditing management systems) positions LPAs within the broader process audit category.

How to implement a Layered Process Audit program (8 steps)

Implementing an LPA program is not a checklist exercise — it is a cultural and operational change. The following eight steps mirror the approach recommended in CQI-8 and refined by mature automotive suppliers.
Step 1 — Secure executive sponsorship
Before any checklists are written, secure an executive sponsor at Layer 4 (ideally the plant manager or operations VP). Their public endorsement of the program, participation in the first Layer 4 audit, and approval of the schedule/resources give the program legitimacy. Without this, Layer 1 and 2 audits degrade into a compliance exercise within 6 months.
Step 2 — Define critical processes
Identify the processes where deviation has the highest risk of causing defects, safety incidents, or customer escapes. Use existing documents — FMEA (Failure Mode and Effects Analysis), Control Plans, Process Flow Diagrams, and turtle diagrams — to surface candidates. Prioritize processes with:
- High severity × occurrence scores in the Process FMEA
- Recent customer complaints or warranty claims
- Known operator skill dependencies
- Regulatory or safety criticality
Step 3 — Build the audit team and assign layers
Constitute an LPA Planning Team (per CQI-8) that includes Quality, Production, Engineering, and a management sponsor. Then assign auditors to each layer:
- Layer 1: Line-level supervisors, team leaders
- Layer 2: Department supervisors, quality engineers, manufacturing engineers
- Layer 3: Plant managers, quality managers
- Layer 4: Executives (plant manager, ops director, VP)
Use a RASIC chart to clarify who is Responsible, who Approves, who Supports, who is Informed, and who is Consulted for each program element.
Step 4 — Develop effective LPA checklists
For each critical process, build a checklist tied to the FMEA/Control Plan failure modes. Questions must be binary (Yes/No), observable, and tied to a specific evidence source. See Writing effective LPA checklist questions below for detailed guidance.
Step 5 — Set the frequency schedule
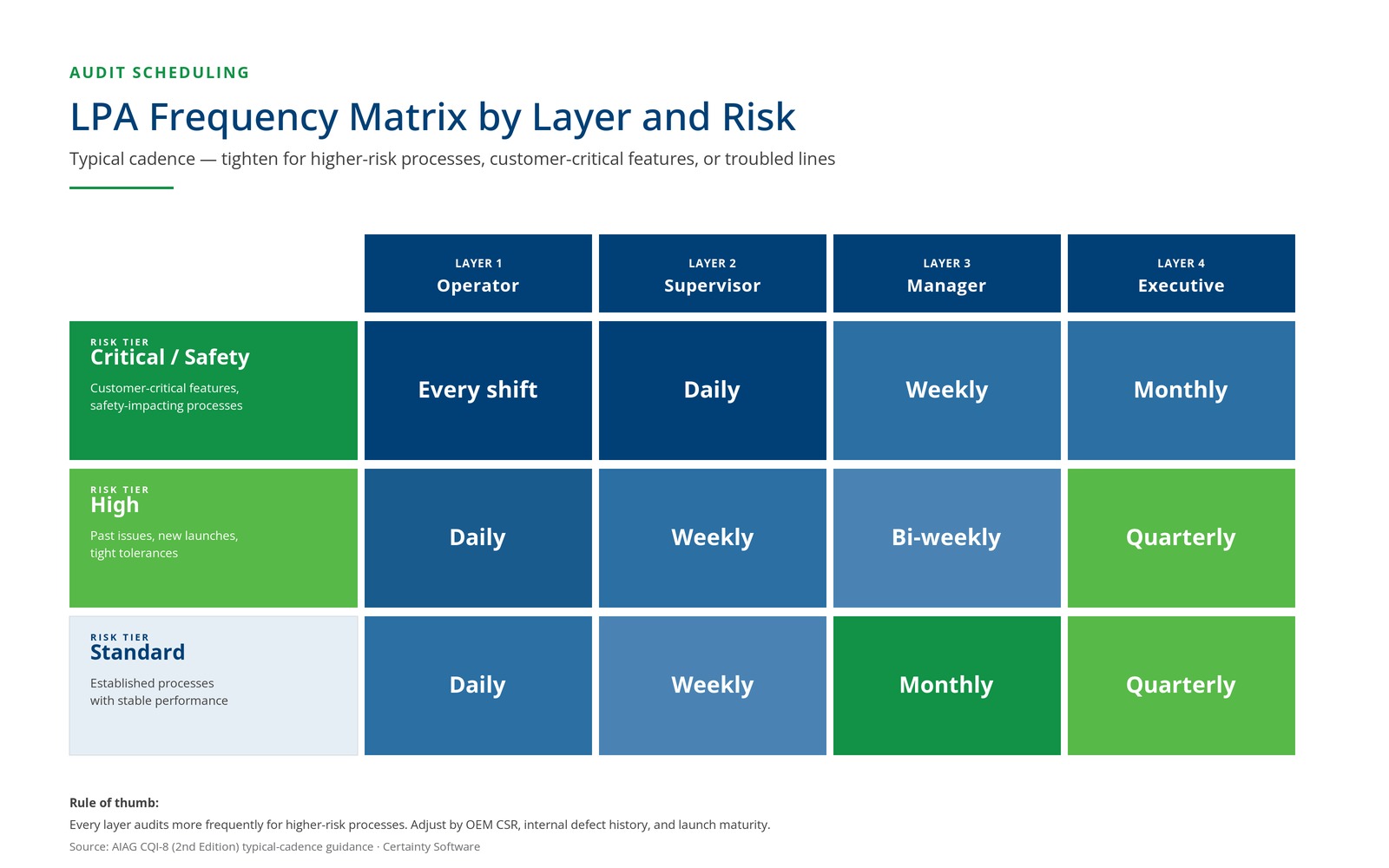
Assign a frequency to each layer and each process. Typical starting points:
- Layer 1: Daily per shift per critical process
- Layer 2: Weekly per critical process
- Layer 3: Monthly per area
- Layer 4: Quarterly per plant (rotating focus)
Adjust for process criticality. Higher-risk processes (new launches, processes with recent non-conformances) warrant tighter frequency.
Step 6 — Train auditors at all layers
Train every auditor on:
- The purpose and philosophy of LPAs (why, not just how)
- The specific checklist they will use (each question, each evidence standard)
- How to escalate findings
- How to avoid leading or coaching the auditee
Layer 4 executives need the same training as Layer 1 operators — perhaps more, because their visible participation models behavior for the entire organization.
Step 7 — Conduct audits and capture evidence
Conduct audits per schedule. At minimum, capture:
- Auditor name, date, time, shift
- Each question’s answer (Yes / No / N/A with justification)
- Evidence (photo, reading, signature, sample)
- Any non-conformance description with immediate containment action
Mobile-first, photo-capable LPA software dramatically improves evidence quality and reduces administrative burden relative to paper checklists.
Step 8 — Close the loop with CAPA
LPA findings must flow into a tracked CAPA workflow with assigned owners, due dates, and verification of effectiveness. Findings that sit in a report binder without action are worse than no LPA program — they document that the organization knew about the issue and did nothing. Use root cause analysis tools (5 Whys, fishbone, 8D) for repeat or severe findings.
Writing effective LPA checklist questions
The quality of an LPA program is determined more by the quality of its questions than by any other factor. Poorly written questions produce false positives, auditor fatigue, and ultimately pencil whipping.
Five rules for effective LPA questions
- Binary and observable. A good LPA question can be answered Yes or No by a single observation. “Is the operator wearing safety glasses?” is observable. “Is the operator working safely?” is not.
- Tied to an evidence standard. Each question should reference a specific evidence source — a work instruction number, a control plan parameter, a torque value, a photo standard. This prevents interpretation drift between auditors.
- Anchored in failure mode risk. Questions should come from Process FMEA failure modes, Control Plan reactions, or documented past non-conformances. Random questions produce random findings.
- Short enough to be done well. Layer 1 checklists should be completable in 5–10 minutes. Beyond that, audit quality declines sharply. If you have more questions than you have minutes, rotate question subsets rather than expanding the checklist.
- Non-leading. Avoid questions that telegraph the expected answer. “Is the work instruction being followed as required by the control plan?” suggests the Yes. “Has the operator performed the first-piece torque check in the last hour?” is neutral.
Examples
| Weak question | Why it fails | Strong rewrite |
|---|---|---|
| “Is the area clean?” | Subjective, no standard | “Are all 5S zones marked and free of items outside their marked location?” |
| “Is the operator following the procedure?” | Not observable | “Has the operator signed the first-off inspection form for the current run?” |
| “Is the machine running correctly?” | Vague | “Does the HMI display the correct recipe number (P/N match) for this part?” |
| “Is safety being followed?” | Scope too broad | “Is the light curtain operational and the safety interlock engaged?” |
Refreshing the checklist
CQI-8 recommends reviewing LPA checklists at least quarterly. Rotate questions out as process learning evolves, rotate new questions in based on recent non-conformances, and retire questions that have a 100% Yes rate over 20+ consecutive audits (they are no longer discriminating).
Preventing pencil whipping and audit fatigue
“Pencil whipping” is the practice of checking every box Yes without actually performing the audit. It is the single most common failure mode of LPA programs, and most organizations significantly underestimate how prevalent it is.
Why pencil whipping happens
- Checklists are too long. Auditors time-pressured into finishing faster than the audit can be honestly performed.
- No consequence for findings, large consequence for missing audits. If “no findings” is rewarded and “missing audits” is punished, auditors optimize for the rewarded behavior.
- Repetitive questions with 100% Yes history. If a question has never been answered No, auditors stop looking.
- No verification mechanism. If nobody ever checks whether the audit actually occurred, it eventually stops occurring.
Six prevention tactics
- Randomize question order. Prevents rote check-the-same-boxes behavior.
- Require photo evidence on a subset of questions. Even one required photo per audit dramatically increases honest completion.
- Set a realistic time window. An audit that “takes” 2 minutes almost certainly isn’t happening — flag any submission under a minimum duration threshold.
- Rotate auditors. Same auditor, same checklist, same station every day = familiarity breeds shortcuts. Rotate assignments monthly.
- Escalate repeat N/As. An auditor answering N/A on the same question repeatedly is signaling the question needs rewriting, not avoidance.
- Conduct periodic “audit the audit” reviews. Layer 3 should spot-check Layer 1 audits — physically revisit stations recently audited and verify the reported condition matches current reality.
Digital LPA software makes these tactics far easier: question randomization, mandatory photo capture, time stamping, GPS geofencing (for multi-site organizations), and audit-the-audit reporting are all native features in purpose-built platforms.

LPA KPIs and benchmarks
LPA programs that are measured improve; programs that aren’t, don’t. CQI-8 2nd Edition specifically emphasizes the integration of KPIs as a core element of program design.
Leading indicators (measure program health)
| KPI | Definition | Typical mature-program benchmark |
|---|---|---|
| Audit completion rate | % of scheduled audits actually performed | ≥ 95% |
| Audit on-time rate | % of audits completed within scheduled window | ≥ 90% |
| Photo/evidence capture rate | % of audits with required evidence attached | ≥ 90% |
| Finding rate by layer | Non-conformances identified per 100 audits | Layer 1: 10–20 / Layer 2: 5–10 / Layer 3: 2–5 |
| Audit duration vs. standard | Actual time vs. expected | Flag audits <50% of expected duration |
Lagging indicators (measure business outcomes)
| KPI | Definition | Typical mature-program benchmark |
|---|---|---|
| CAPA closure cycle time | Median days from LPA finding to verified closure | ≤ 14 days for routine, ≤ 5 days for safety-critical |
| Repeat issue rate | % of findings that recur at the same station within 90 days | ≤ 10% |
| First-pass yield (FPY) | % of units produced right the first time | Trend should improve year over year |
| Cost of Poor Quality (CoPQ) % of sales | Scrap + rework + warranty + inspection + field failure cost | Mature program target: below 5% (from 15–20% baseline) |
| Customer PPM (parts per million defective) | Customer-reported defects per million shipped | Continuous downward trend |
A finding rate of zero at Layer 1 is not a sign of a perfect process — it is almost always a sign of a broken audit. Expect and encourage findings; the program’s purpose is to surface them, not to hide them.
See our continuous improvement assessment guide for broader KPI framing.
LPAs beyond automotive
Though CQI-8 and LPAs originated in automotive, the methodology translates well to any industry where consistent process execution determines quality and safety outcomes.
Aerospace (AS9100)
Aerospace manufacturers use LPA-style process audits to satisfy AS9100 Clause 9.2 internal audit requirements and manage the critical-to-quality parameters (Key Characteristics) required for flight-safety parts. Frequency is often tighter; evidence requirements (photo, measurement, inspection stamp) are stricter.
Medical devices (ISO 13485)
Medical device manufacturers use LPA methodologies to support ISO 13485 process validation and 21 CFR Part 820 Quality System Regulation requirements. Audit questions frequently tie to Design History File, Device Master Record, and Device History Record elements.
Food and beverage (HACCP, SQF, BRCGS)
Food manufacturers use LPA-style audits to verify HACCP Critical Control Points, allergen control, sanitation preconditions, and traceability practices. These are often branded as “CCP verification audits” or “pre-op inspections” but follow the LPA pattern of multi-layer, structured, frequent verification.
Pharmaceuticals (cGMP)
Pharmaceutical manufacturers use LPAs to verify cGMP compliance at the point of operation, complementing formal internal audits and FDA readiness programs.
General manufacturing and service
Any organization whose quality depends on consistent process execution can benefit from LPAs. CQI-8 2nd Edition explicitly expands guidance for non-manufacturing applications — engineering change processes, purchasing, order fulfillment, and service delivery processes are increasingly audited using LPA-style methods.
LPA software: what to look for
Purpose-built LPA software is now the standard approach for organizations serious about program rigor. Paper-based LPAs survive only in small operations or as transitional tooling.
Eight capabilities that separate serious platforms from form-builder tools
- Layered scheduling. Different frequencies, different checklists, different auditors per layer, with automated assignment and reminders.
- Mobile-first data capture. The audit must happen at the station, not back at a desk. Offline capability for shop-floor connectivity gaps.
- Mandatory photo / evidence capture. Required on specific questions, time-stamped, geo-located.
- Automated CAPA workflow. Non-conformances flow into an assignment → due date → verification loop without manual rekeying.
- Question randomization and rotation. Prevent rote answering.
- Dashboards and trend analytics. Finding rate by station, layer, question, auditor. Drill-down to specific audits.
- Integration with FMEA, Control Plan, and QMS. So checklist questions link back to the source-of-truth documents.
- Audit-the-audit reporting. Layer 3/4 can see what was audited and spot-check veracity.
See our 10 things to consider when selecting LPA software for a detailed buyer’s guide.
See how Certainty supports Layered Process Audits
Mobile, configurable LPA platform · Layered scheduling · Mandatory photo evidence · Automated CAPA · Real-time dashboards
Book a DemoFrequently asked questions
What is a Layered Process Audit?
A Layered Process Audit (LPA) is a structured, multi-level auditing method where operators, supervisors, managers, and executives each conduct frequent, standardized audits of critical processes using pre-defined checklists. LPAs verify process inputs and behaviors — not finished products — to catch deviations before they produce defects.
What is the purpose of a Layered Process Audit?
The purpose of an LPA is to proactively prevent defects by verifying that critical process parameters are being controlled consistently at the point of work. Because multiple organizational layers participate, LPAs also drive management engagement with quality and produce the audit evidence required by ISO 9001, IATF 16949, and OEM Customer Specific Requirements.
Are Layered Process Audits required by IATF 16949?
Not directly. IATF 16949:2016 Clause 9.2.2.3 requires manufacturing process audits but does not specify LPAs as the method. However, OEM Customer Specific Requirements — notably General Motors and Stellantis — do require LPAs per the AIAG CQI-8 guideline. Most automotive suppliers therefore treat LPAs as effectively required.
How often should Layered Process Audits be conducted?
Frequency is assigned by layer. A typical schedule is: Layer 1 (operators) daily or per shift; Layer 2 (supervisors) weekly; Layer 3 (managers) monthly; Layer 4 (executives) quarterly. Higher-risk processes — recently launched, recently non-conforming, or safety-critical — warrant tighter frequency at every layer.
What is the difference between a Layered Process Audit and a Gemba walk?
LPAs are structured, checklist-driven, and multi-layered; findings flow into a formal CAPA system. Gemba walks are observational, open-ended, and focused on understanding how work actually happens rather than verifying compliance. Both are valuable and complementary — see the comparison table above.
What is CQI-8 and is it mandatory?
CQI-8 is the Layered Process Audit Guideline published by the Automotive Industry Action Group (AIAG). It is a guideline, not a mandatory standard. However, OEM Customer Specific Requirements from major automakers reference CQI-8 directly, effectively making it mandatory for their suppliers.
What KPIs should we measure for our LPA program?
Measure leading indicators (audit completion rate, on-time rate, evidence capture rate, finding rate by layer) and lagging indicators (CAPA closure cycle time, repeat issue rate, first-pass yield, Cost of Poor Quality). Target ≥95% completion, ≥90% on-time, and a downward trend on CoPQ and repeat issues.
How do you prevent pencil whipping in LPAs?
Randomize question order, require photo evidence on key questions, set minimum audit duration thresholds, rotate auditor assignments, escalate repeat N/A responses, and conduct “audit the audit” verifications where higher layers spot-check completed audits against current conditions. Digital LPA platforms automate most of these controls.
Related resources
Quality standards and audit foundations
- IATF 16949: Automotive Quality Management Standard
- What is the ISO 9001 Audit?
- ISO 19011: A Comprehensive Guide to Quality Management Auditing
- What is VDA 6.3? (Automotive Process Audit Standard)
LPA and continuous improvement practices
- Gemba Walk: The First Step Toward Continuous Improvement
- The 4 Phases of a Continuous Improvement Assessment
- Kaizen for Continuous Improvement
- What is 6S Lean?
Problem-solving and CAPA
- CAPA Process: 6 Steps for Improved Quality Management
- A Comprehensive Guide to Root Cause Analysis
- 8D Problem Solving: A Guide for Businesses
- Implementing DMAIC Process: A Guide
- 4 Ways You Can Prevent Non-Conformances
Tools, checklists, and product resources
- LPA Whitepaper: 2023 Guide to Layered Process Audits
- Poka Yoke: The Ultimate Guide to Error Prevention
- Certainty LPA Use Case
- Quality Audit Software Overview



